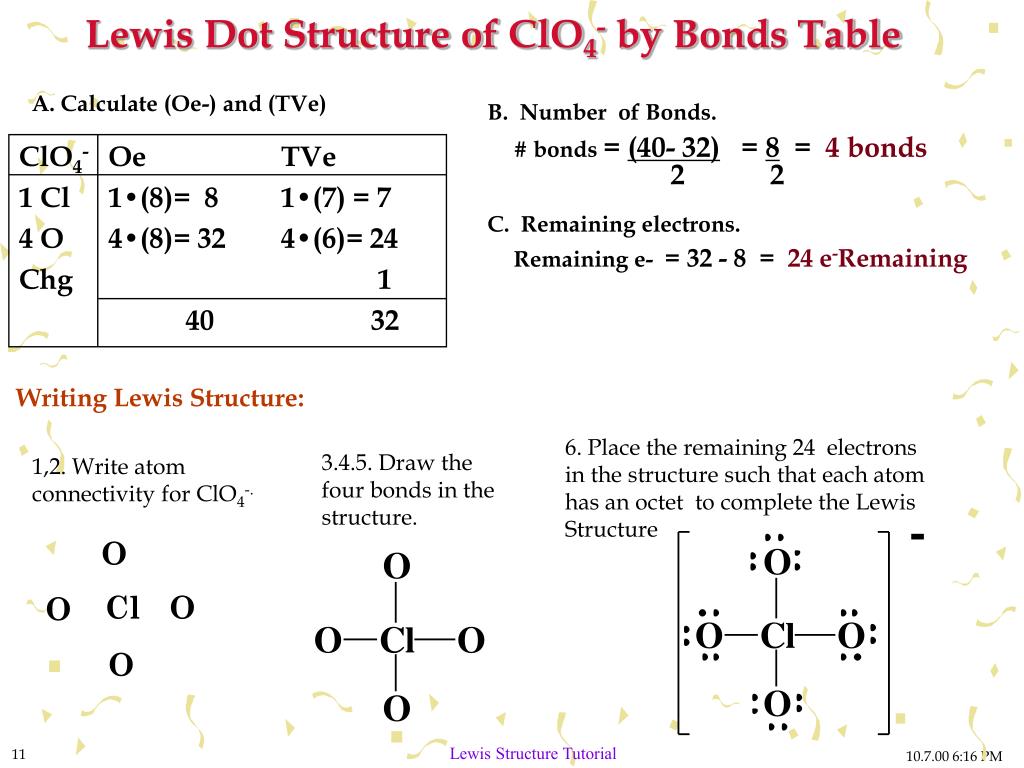
Draw Lewis Structure
FCN lewis structure, also known by the chemical name as cyanogen fluoride, is an inorganic molecule with a molecular weight of 45.016 g/mol. Some facts about FCN lewis structure : Molar mass/Molecular weight = 45.016 g/mol. Boiling point = -46.17 0 C , Melting point = - 82 0 C .

C2h2br2 Nonpolar Lewis Structure
Strong electron delocalization in your best Lewis structure will also show up as donor-acceptor interactions. Interactions greater than 20 kJ/mol for bonding and lone pair orbitals are listed below. The interaction of lone pair donor orbital, 8, for F1 with the antibonding acceptor orbital, 70, for N2-C3 is 70.3 kJ/mol.

FCN Lewis Structure How to Draw the Lewis Dot Structure for FCN
8.6: Resonance Structures is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. Some molecules have two or more chemically equivalent Lewis electron structures, called resonance structures. Resonance is a mental exercise and method within the Valence Bond Theory of bonding that..
:max_bytes(150000):strip_icc()/ScreenShot2018-11-19at11.40.52PM-5bf3909a46e0fb00510dbd6d.png)
Lewis Structure Definition and Example
A step-by-step explanation of how to draw the CN- Lewis Dot Structure (Cyanide ion).Note: there should be brackets around the final Lewis Structure with a -1.

Fcn Lewis Dot Structure Abbreviations Fch Family Cohesion Fcn Family
Choose the best Lewis structure for FCN and explain why it is the best C==N: 6_C=n: Structure is better because it is symmetrical with 2 double bonds Structure b is better because it has all formal charges that are zero while structure has a + | charge on the N and a - ] charge on the F
/Lewis-dot-structure-58e5390f3df78c5162b4c3db.jpg)
Best How To Draw The Lewis Dot Structure in the year 2023 The ultimate
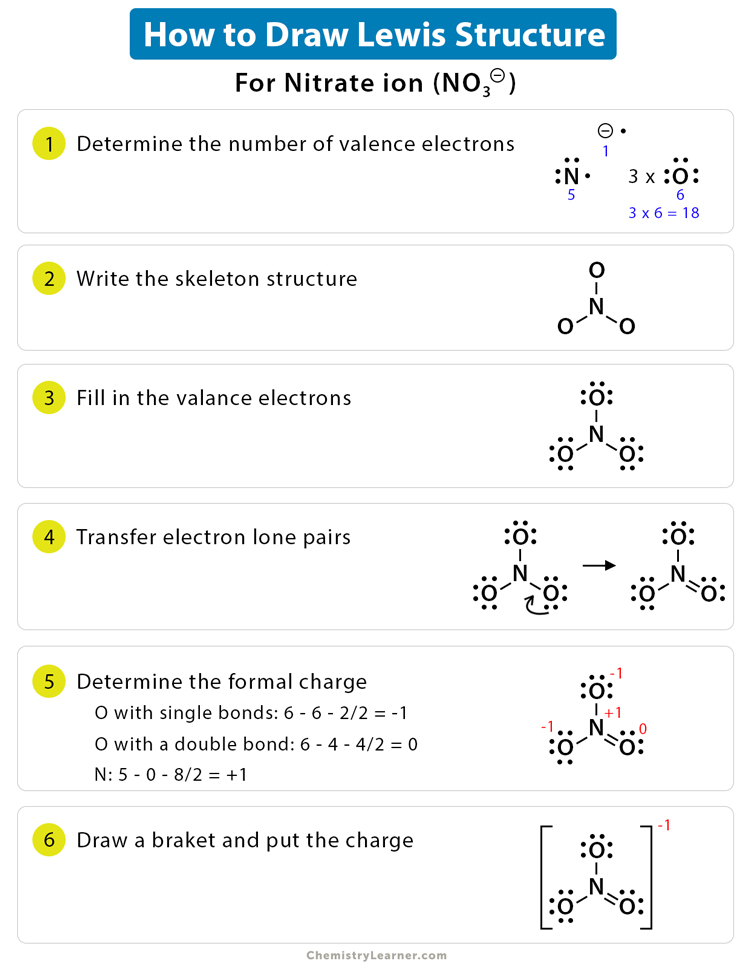
About. Transcript. We can draw Lewis structures for polyatomic ions (ions containing multiple atoms) using the same stepwise procedure as for neutral molecules. In this video, we'll see how to construct the Lewis diagram of the cyanide ion (CN⁻). Created by Sal Khan.
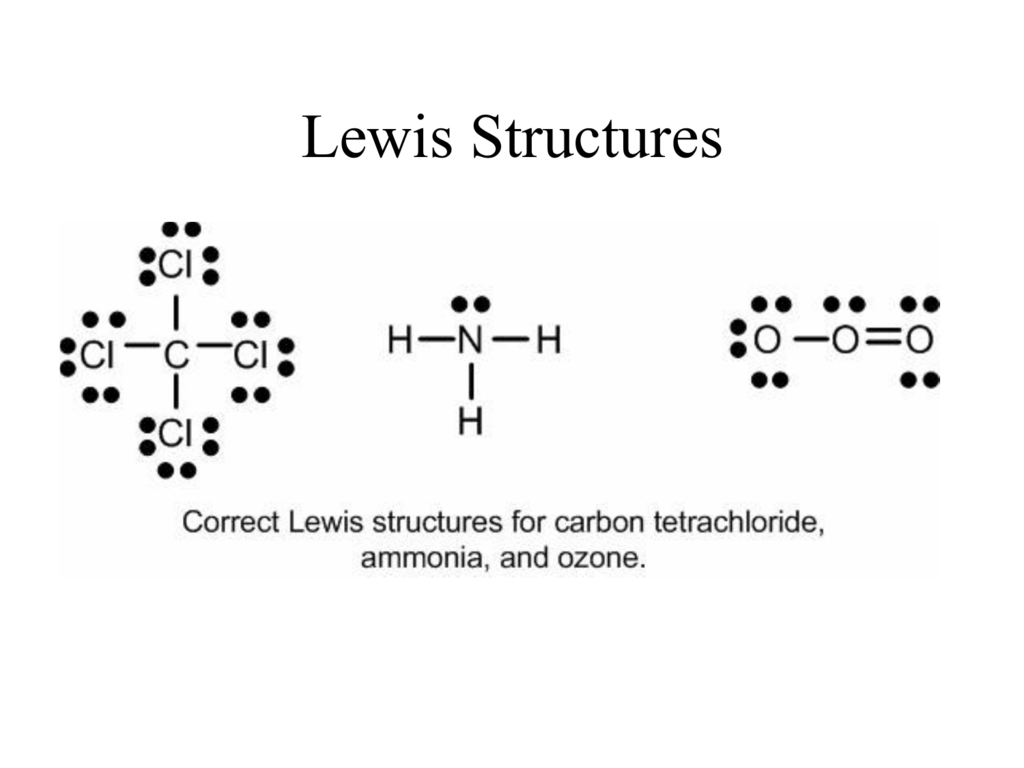
Breathtaking Tips About How To Draw A Lewis Dot Structure Ballchicken
Cyanogen fluoride (molecular formula: FCN; IUPAC name: carbononitridic fluoride) is an inorganic linear compound which consists of a fluorine in a single bond with carbon, and a nitrogen in a triple bond with carbon. It is a toxic and explosive gas at room temperature. It is used in organic synthesis and can be produced by pyrolysis of cyanuric fluoride or by fluorination of cyanogen.
lewis dot structure chart Focus
What is the Lewis structure of [//substance:FCN//]? Natural Language; Math Input; Extended Keyboard Examples Upload Random. Compute answers using Wolfram's breakthrough technology & knowledgebase, relied on by millions of students & professionals. For math, science, nutrition, history, geography, engineering, mathematics, linguistics, sports.

How To Draw Lewis Structures In Chemdraw Images and Photos finder
Summary. Following the Octet Rule for Lewis Dot Structures leads to the most accurate depictions of stable molecular and atomic structures and because of this we always want to use the octet rule when drawing Lewis Dot Structures. There are three exceptions: (1) When there are an odd number of valence electrons, (2) When there are too few.
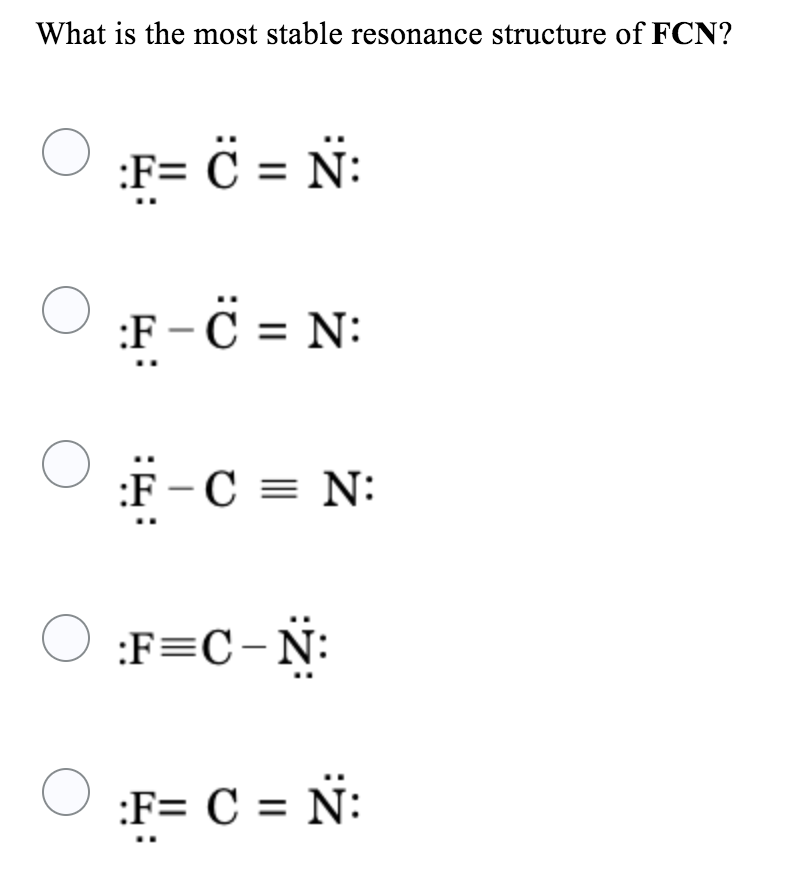
Solved What is the most stable resonance structure of FCN?
In FCN Lewis structure, there is a single bond between carbon and fluorine atom, and a triple bond between carbon and nitrogen atom. The fluorine atom has three lone pairs, and the nitrogen atom has one lone pair. Contents. Steps #1 First draw a rough sketch

draw the main lewis structure of nofnof. darnelllemmings
FCN is a chemical formula for cyanogen fluoride. And to help you understand the Lewis Structure of this molecule, we are going to share our step-by-step met.
[Solved] 1. Draw Lewis structures for each of the following. Use VSEPR
6 Steps to Draw the Lewis Structure of FCN Step #1: Calculate the total number of valence electrons. Here, the given molecule is FCN. In order to draw the lewis structure of FCN, first of all you have to find the total number of valence electrons present in the FCN molecule. (Valence electrons are the number of electrons present in the outermost shell of an atom).

How to Draw Lewis Structures RodneytaroBall
Steps of drawing FCN lewis structure Step 1: Find the total valence electrons in FCN molecule. In order to find the total valence electrons in an FCN molecule, first of all you should know the valence electrons present in fluorine atom, carbon atom as well as nitrogen atom. (Valence electrons are the electrons that are present in the outermost orbit of any atom.)
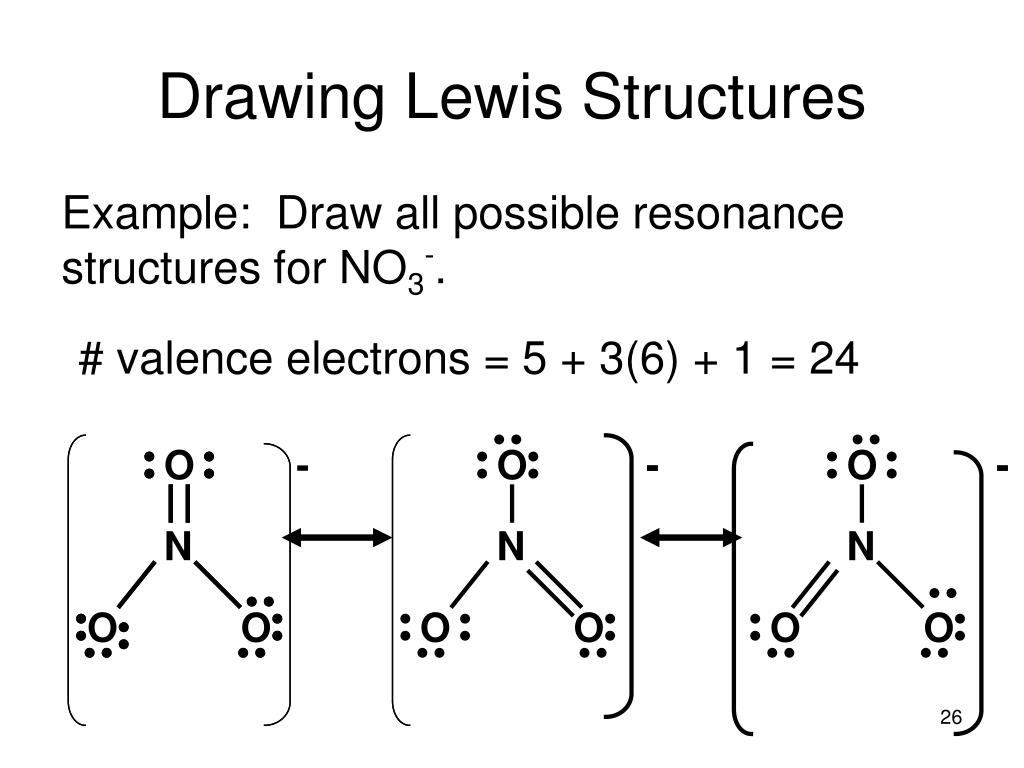
Draw Lewis Structure
FCN Lewis Structure FCN is a chemical formula for cyanogen flouride. And to help you understand the Lewis Structure of this molecule, we are going to.
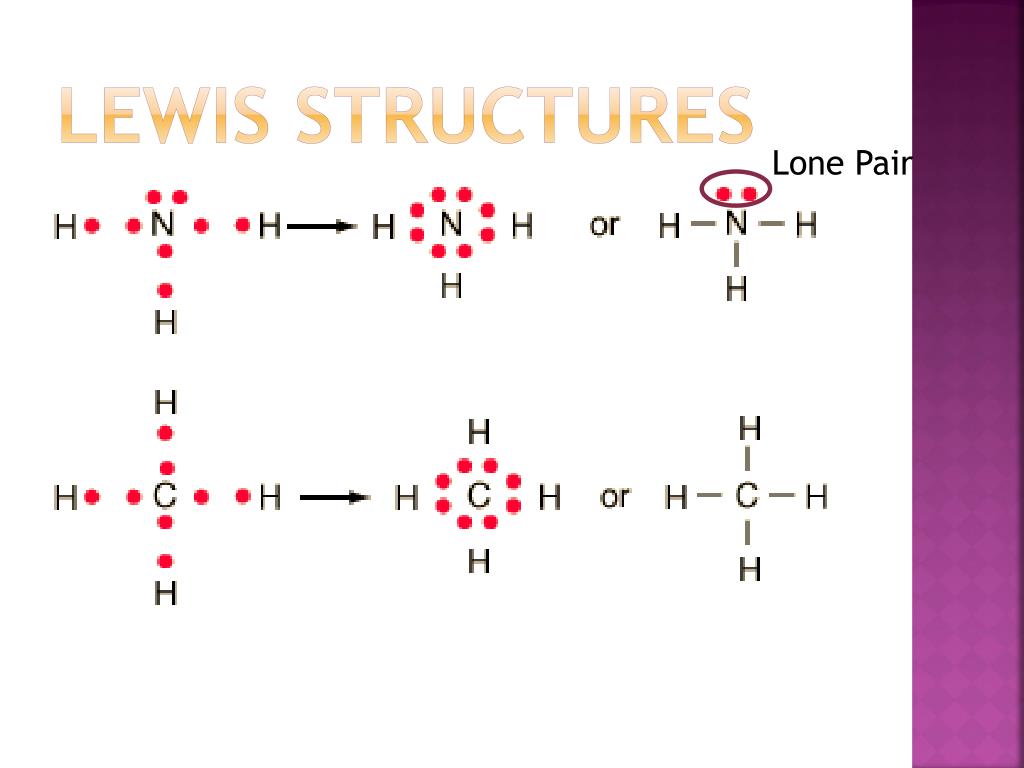
Draw Lewis Structure
Step 1: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. When drawing the structure of an ion, be sure to add/subtract electrons to account for the charge. Step 2: Connect the atoms to each other with single bonds to form a "skeleton structure.".
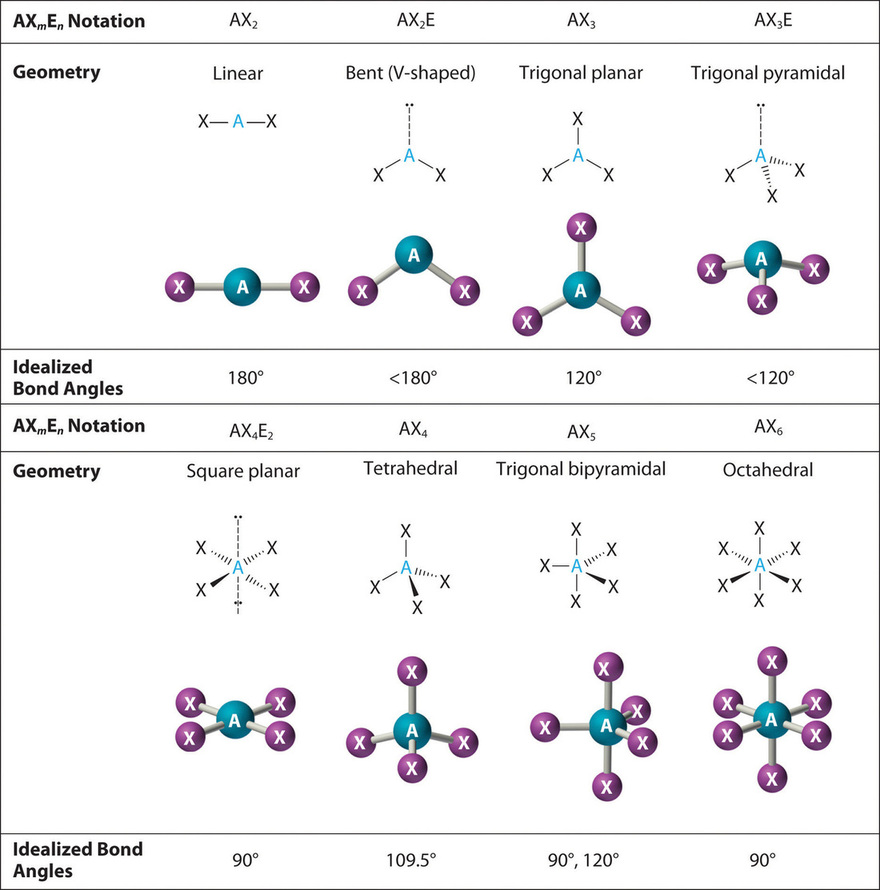
The VSEPR Model
The former, known as a 'Lewis dot diagram,' indicates a pair of shared electrons between the atomic symbols, while the latter, known as a 'Lewis structure,' uses a dash to indicate the pair of shared electrons that form a covalent bond. More complicated molecules are depicted this way as well.